The Cost of Oversimplified Biology
Despite decades of advances in molecular biology and drug discovery, the reality remains sobering: over 90% of drug candidates that succeed in preclinical testing ultimately fail in clinical trials. At the heart of this issue lies a simple but often overlooked concept: traditional in vitro assays are built around a simplified view of biology.
Cells are cultured on rigid plastic substrates and exposed to drugs in environments that are mechanically inert. These models, although easy and cost-effective, lack the physical context in which these cells naturally exist. There is no fluid flow, no cyclic stretching, no compression, none of the dynamic forces that cells experience in living tissues. For decades, these static assays have served as the backbone of biological research but this simplicity comes at the cost of losing physiological reality.
Cells Are Mechanical Systems
Cells are not passive building blocks; they are dynamic entities constantly responding to an environment full of mechanical cues. Through mechanotransduction, they sense stiffness from the extracellular matrix (ECM), fluid shear from blood flow, tension from tissue stretching and many other cues that reshape cytoskeletal organization, alter gene expression and dictate cell fate1.
These mechanical inputs are integral to maintaining tissue homeostasis, cellular function and play a fundamental role in disease pathology. For example, rhythmic stretching from breathing in the lungs preserves epithelial barrier function and alveolus architecture, but during fibrosis, altered mechanical properties trigger pathological programs such as epithelial–mesenchymal transition and fibrotic remodelling2.Similarly, in healthy vasculature, shear stress and cyclic strain from blood flow help maintain endothelial alignment and vessel tone, whereas disrupted flow and vessel stiffening promote maladaptive remodelling and favour diseaseprogression3. In tumour microenvironments, malignant cells exploit mechanical cues to promote invasion, gene reprogramming, and therapy resistance through increased matrix stiffness, compressive stress, and fluid shear4.
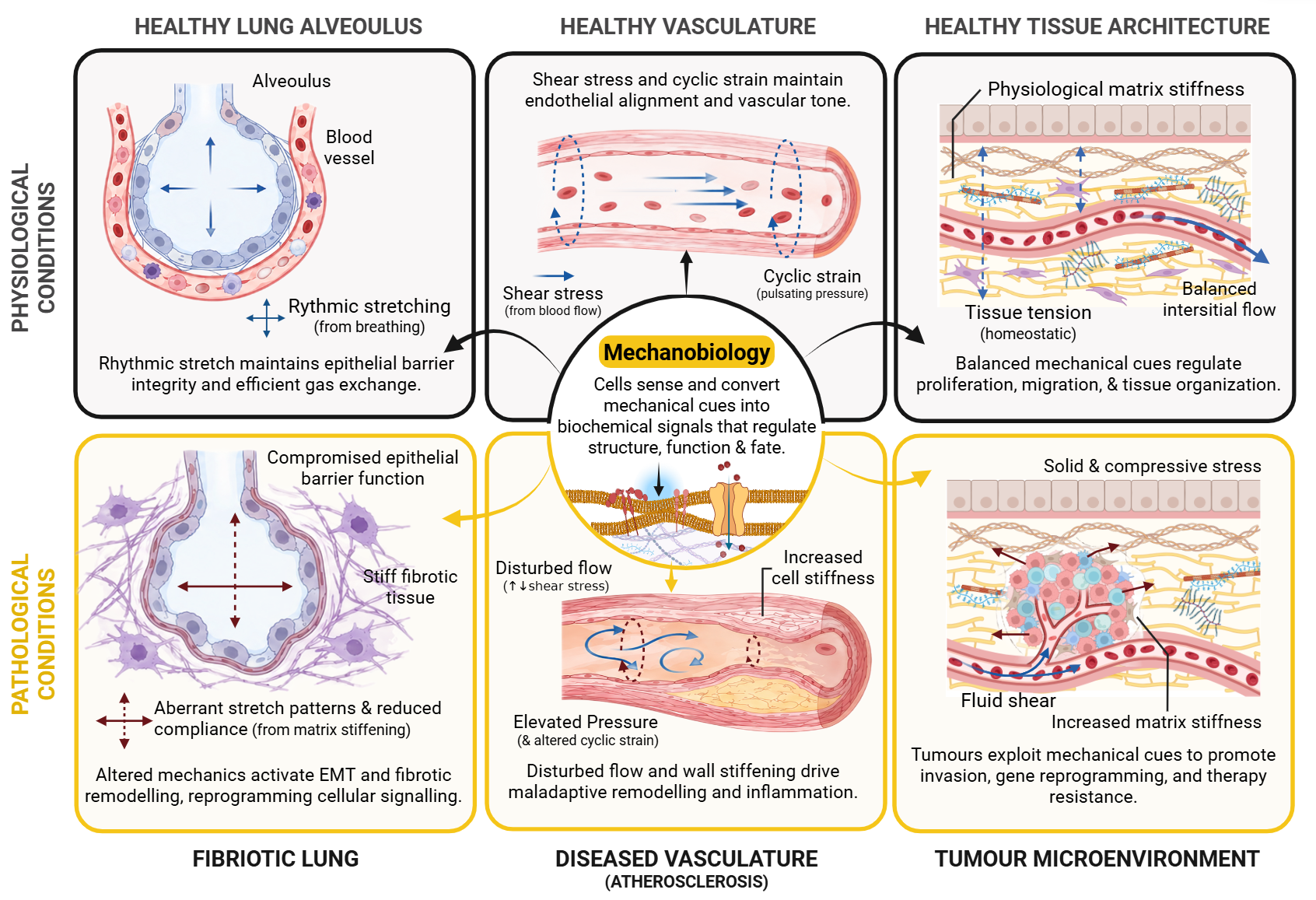
From Simplification to Misprediction: Translational Failure
When cells are removed from their mechanical context and placed in static environments, their behaviour changes drastically and their phenotype no longer reflects their in vivo counterpart.
This mismatch helps explain why preclinical success so often fails to translate into clinical benefit. A compound may look effective in a simplified assay yet perform poorly once it encounters the complexity of human tissue. Mechanical context is one hidden variable that can shape predictability, efficacy and toxicity. This contributes to the well-known problem of limited translational predictivity in preclinical research5. When models leave out the relevant physical environment, they can create early confidence that later turns into costly failure.
Bridging the Gap: Reintroducing Mechanical Reality
As a result, the field is experiencing a shift towards mechanosensitive biology, placing biomechanics at the core of the experimental design rather than an afterthought.
A growing class of technologies that incorporate mechanical forces into in vitro systems have been developed. Technologies such as microfluidic devices, mechanical stretchers and organ-on-chip platforms allow researchers to better recreate the cellular microenvironment. These systems can introduce controlled fluid flow, cyclic stretching or modulate substrate stiffness, and thus provide a more complete picture of cellular behaviour.
At 60Nd, our goal is to help bridge the gap between simplified in vitro models and the mechanical complexity of living systems. We have developed NeoMag6, a programmable mechanical actuation platform designed to recreate user-defined physiological and pathological conditions in vitro. The system allows precise, contact-free control of key physical parameters, with all stimulation programmed remotely and seamlessly integrated into standard lab workflows. Its portable design and 96-well compatible dimensions make it adaptable to common imaging systems, incubators, and experimental setups.
With NeoMag, researchers can tune the cellular microenvironment, modulating substrate stiffness to that of different tissue types (from soft brain to rigid fibrotic tissue) and apply sustained or cyclic forces such as stretch, compression, or shear stress.

These models are more informative and thus lead to better predictions of disease pathology, drug efficacy and safety. It is important to note that the goal is not to replace existing methods but to complement and enhance them. Static assays will continue to play a role in early-stage research. However, as drug candidates progress, integrating mechanical context may become essential for reducing attrition costs and improving success rates.
Looking Ahead
If you’d like to explore more about how mechanobiology is reshaping preclinical research, stay tuned for our next blog. For more information on NeoMag, you can learn more here.
At 60Nd, we’re working toward a future where the physical and biochemical dimensions of life are studied together, not in isolation.
Reference List
1. Humphrey JD, Dufresne ER, Schwartz MA. Mechanotransduction and extracellular matrix homeostasis. Nature Reviews Molecular Cell Biology. 2014 Oct 22;15(12):802–12.doi:10.1038/nrm3896
2. Link PA, Tschumperlin DJ. Mechanobiology and Resolution of Lung Fibrosis. Annual Review of Physiology. 2026 Feb 10;88(1):487–511.doi:10.1146/annurev-physiol-031725-021041
3. Humphrey JD, Schwartz MA. Vascular Mechanobiology: Homeostasis, adaptation, and disease. Annual Review of Biomedical Engineering. 2021 Jul 13;23(1):1–27.doi:10.1146/annurev-bioeng-092419-060810
4. Agrawal A, Javanmardi Y, Watson SA, Serwinski B, Djordjevic B, Li W, et al. Mechanical signatures in cancer metastasis. npj Biological Physics and Mechanics. 2025 Feb4;2(1). doi:10.1038/s44341-024-00007-x
5. Loewa A, FengJJ, Hedtrich S. Author correction: Human disease models in drug development. Nature Reviews Bioengineering. 2023 Jun 13;1(8):606–606.doi:10.1038/s44222-023-00088-8
6. Gomez‐Cruz C, Fernandez‐de la Torre M, Lachowski D, Prados‐de‐Haro M, del Río Hernández AE, Perea G, et al. Mechanical and functional responses in astrocytes under alternating deformation modes using magneto‐active substrates. Advanced Materials. 2024 Apr21;36(26). doi:10.1002/adma.202312497